graph TD CAD["Coronary Artery Disease (CAD)"] StableAngina["Chronic Stable Angina"] ACS["Acute Coronary Syndrome (ACS)"] UnstableAngina["Unstable Angina"] NSTEMI["NSTEMI"] STEMI["STEMI"] Variant["Variant (Prinzmetal) Angina"] CAD --> StableAngina CAD --> ACS CAD --> Variant ACS --> UnstableAngina ACS --> NSTEMI ACS --> STEMI
Etiology
Pathophysiology
Possibilities
- Left anterior descending artery (45%): infarction of the anterior wall and anterior septum of the LV
- Right coronary artery: infarction of the posterior wall, posterior septum, and papillary muscles** of the LV
- Left circumflex artery: infarction of the lateral wall of the LV
LV vs RV
| Feature | Left Ventricle | Right Ventricle |
|---|---|---|
| Muscle mass | High | Low |
| Resting oxygen extraction | High | Low |
| Coronary perfusion | During diastole only | Throughout cardiac cycle |
| Collateral circulation | Less developed | More developed |
| Ischemic preconditioning | Low | High |
The relatively low systolic pressure of the RV (eg, ≤25 mm Hg) allows for coronary perfusion throughout the cardiac cycle
Type I vs type II
| Feature | Type 1 MI | Type 2 MI |
|---|---|---|
| Cause | Plaque rupture/erosion → thrombus | Myocardial oxygen supply < demand without acute atherothrombosis |
| Triggers | ACS | Sepsis, anemia/bleeding, hypoxia, arrhythmia, hypo-/HTN, PE |
| ECG | STEMI or NSTEMI ischemic changes common | Ischemic or nonspecific changes; STEMI uncommon (except spasm) |
| Troponin | Rise/fall; often larger (variable) | Rise/fall; often smaller but can be large (depends on severity) |
| Treatment | ACS meds ± urgent PCI | Fix trigger; CAD/antithrombotics/PCI case-by-case |
Clinical features
Tip
The standard 12-lead ECG is essentially designed to look at the Left Ventricle. So these positions of the heart (such as anterior, inferior, etc.) all refer to the wall of the left ventricle.
- Anterior/Septal (LAD; V1–V4)
- More HF/pulm edema, hypotension (large infarct)
- VT/VF risk; VSD (3–5 d)
- Inferior (RCA>>LCX; II, III, aVF)
- More N/V, epigastric pain
- Bradycardia, AV block (Mobitz I)
- Papillary muscle rupture → acute MR (2–7 d) c
- Posteromedial papillary muscle is usually supplied by PDA (RCA in most people) and has single blood supply.
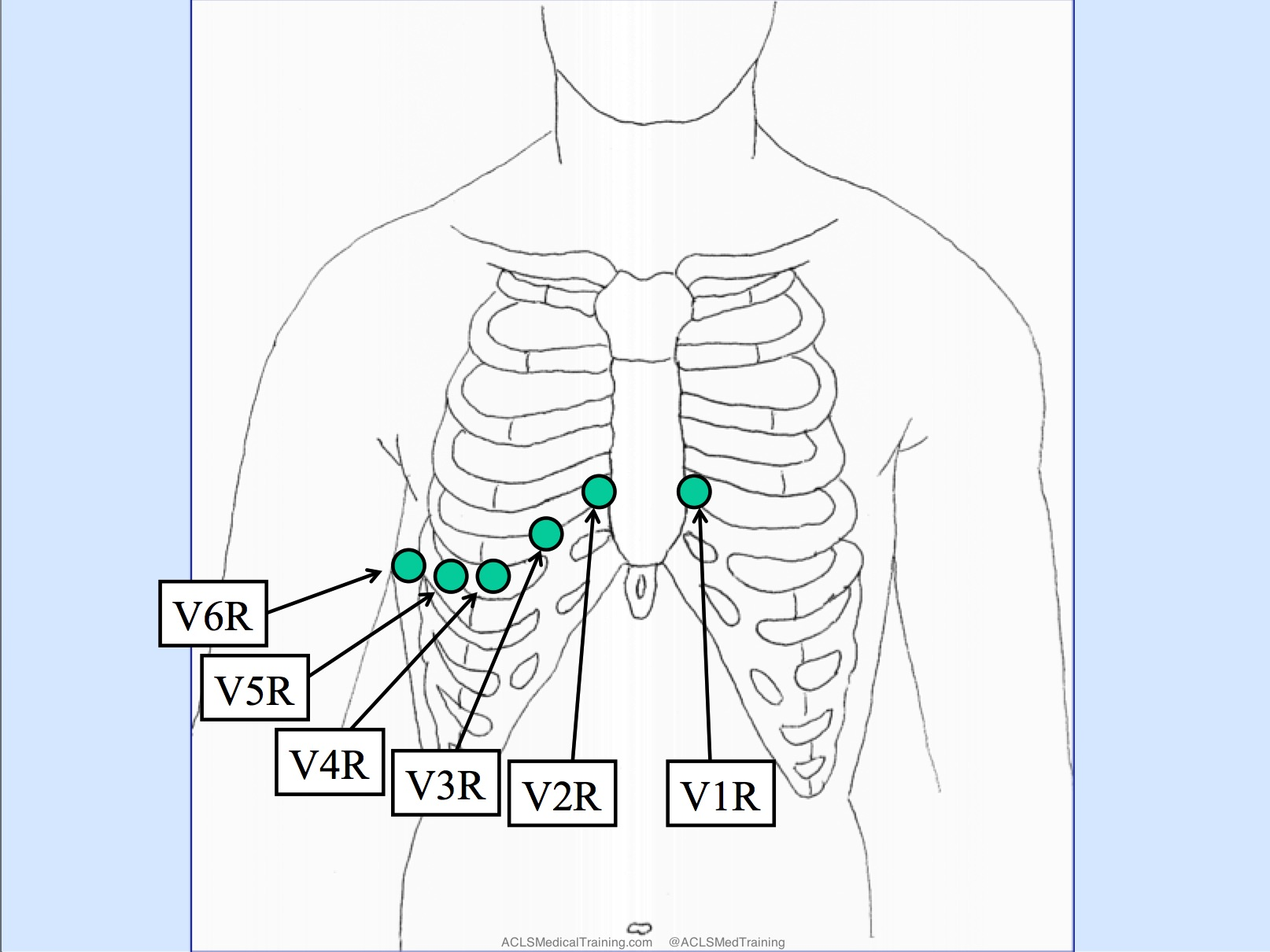
- Right ventricular (prox RCA; V4R)
- Hypotension + JVD + clear lungs (preload-dependent)
- Avoid nitrates/diuretics, give IVF
- Lateral (LCX/diagonal; I, aVL, V5–V6)
- Typical angina; ± HF if large
- Posterior (LCX/RCA; V7–V9)
- ST↓ + tall R in V1–V3; confirm V7–V9 ST↑
Diagnostics
- Best Initial Test: ECG (Do within 10 mins).
- Serial ECGs should be performed (at baseline, then every 15–30 minutes initially) to identify progression to MI. c
- Most Specific Test: Cardiac Troponin I or T.
- Rises in 3-4 hours; peaks at 24 hours.
- Step 2 Tip: If initial troponin is negative but suspicion is high, repeat in 2-3 hours.
- CXR: To rule out aortic dissection (widened mediastinum) if presentation is tearing pain.
Pathology
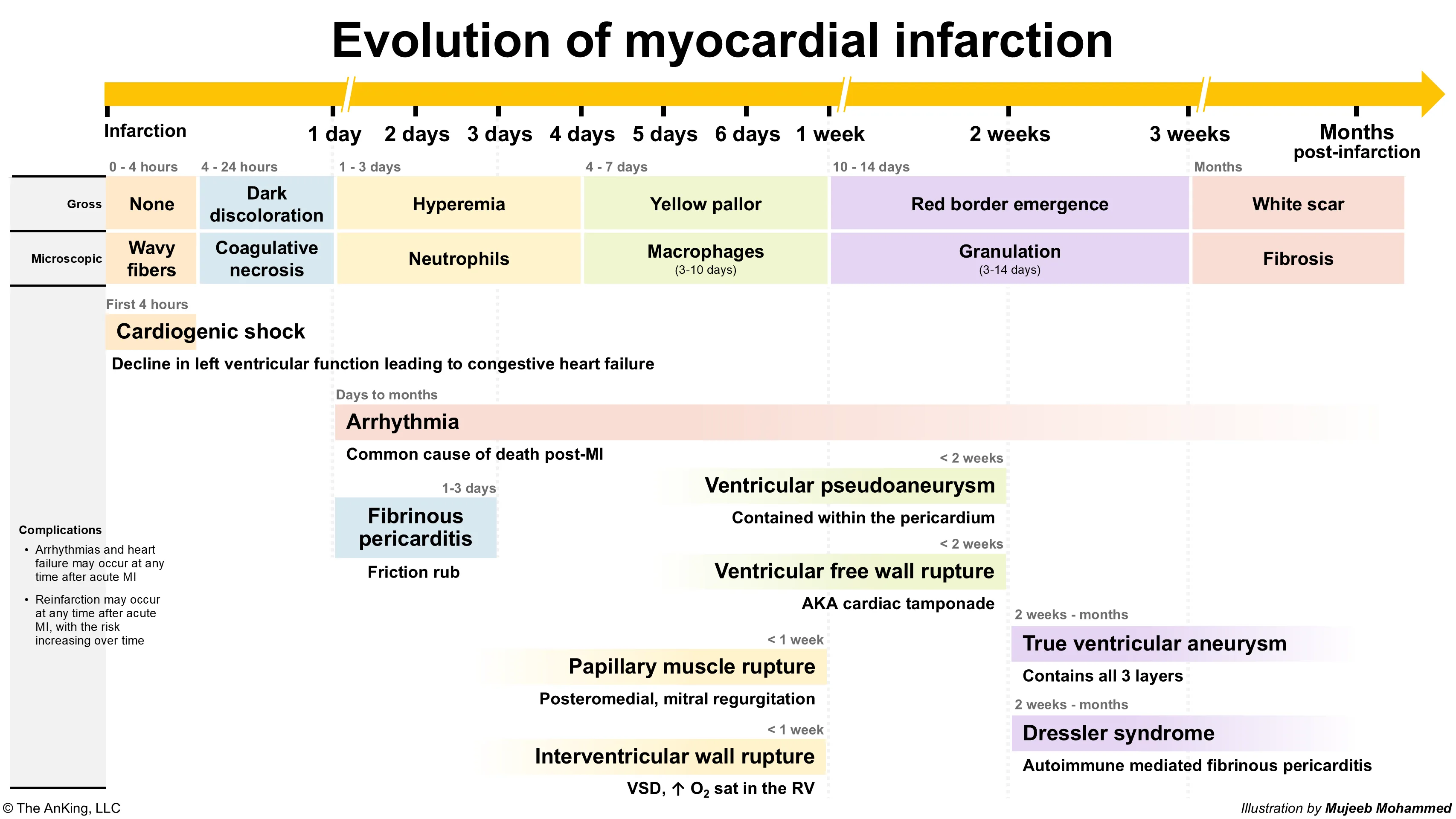
| Time After Myocardial Infarction | Predominant Light Microscopic Changes |
|---|---|
| 0-4 hours | No visible change t |
| 4-12 hours | Wavy fibers with narrow, elongated myocytes |
| 12-24 hours | Myocyte hypereosinophilia with pyknotic (shrunken) nuclei |
| 1-3 days | Coagulation necrosis (loss of nuclei & striations) Prominent neutrophilic infiltrate |
| 3-7 days | Disintegration of dead neutrophils & myofibers Macrophage infiltration at border areas |
| 7-10 days | Robust phagocytosis of dead cells by macrophages Beginning formation of granulation tissue at margins |
| 10-14 days | Well-developed granulation tissue with neovascularization |
| 2-8 weeks | Progressive collagen deposition & scar formation |
Loss of cardiomyocyte contractility occurs within 60 seconds after the onset of total ischemia. When ischemia lasts less than 30 minutes, restoration of blood flow leads to reversible contractile dysfunction (myocardial stunning), with contractility gradually returning to normal over the next several hours to days. However, after about 30 minutes of total ischemia, ischemic injury becomes irreversible. t
Treatment
Acute Coronary Syndrome (ACS: UA, NSTEMI, STEMI)
- Immediate Stabilization (All ACS):
- ABC, IV access, Cardiac Monitor.
- Aspirin: 325mg chewable (loading dose) - Mortality benefit.
- P2Y12 Inhibitor: Clopidogrel/Ticagrelor (DAPT).
- Anticoagulation: Unfractionated Heparin (esp if PCI planned) or LMWH. c
- Nitroglycerin: SL or IV for pain/HTN (Avoid in Right Ventricular MI - II, III, aVF; preload dependent). c
- Beta-Blocker: Metoprolol (within 24h if stable, no HF/bradycardia).
- Statin: High-intensity (Atorvastatin 80mg).
- Reperfusion Strategy (STEMI):
- PCI (Gold Std): Door-to-balloon <90 min (or <120 min if transferrequired).
- Fibrinolysis (tPA): If PCI unavailable within 120 min. Door-to-needle <30 min.
- NSTEMI/UA Strategy:
- TIMI/GRACE Score: Risk stratify.
- Early Invasive (Angio): Refractory angina, hemodynamic instability, new HF, high risk score.
- Ischemia-Guided: Medical mgmt, stress test prior to discharge.
Right Ventricular Infarct (Inferior Wall MI)
Link to original
- Clue: ST elevation in II, III, aVF. Hypotension, Clear lungs, JVD.
- Next Step: Get Right-sided ECG (look for ST elevation in V4R).
- Management: IV Fluids (maximize preload) c . Avoid Nitrates/Diuretics (they drop preload).
- ↑ RV preload (more stretch on RV myocytes)
- Via Frank-Starling law, even a dysfunctional RV can generate slightly more contractile force with increased filling
- This pushes more blood through the pulmonary circulation → ↑ LV filling → ↑ CO
Arrythmia
Class IB antiarrhythmics treat ventricular arrhythmias, especially in ischemic tissue (e.g. post-MI)
Note
Ischemia leads to slow cellular depolarization that inactivates sodium channels, and therefore enhanced binding of IB drugs.
Complications
| Complication | Time course | Clinical findings |
|---|---|---|
| Papillary muscle rupture/dysfunction* | Acute or within 3-5 days | Severe pulmonary edema, respiratory distress New early systolic murmur (acute MR) Hypotension/cardiogenic shock |
| Interventricular septum rupture | Acute or within 3-5 days | Chest pain New holosystolic murmur Hypotension/cardiogenic shock Step up in O2 level from RA to RV |
| Free wall rupture** | Within 5 days or up to 2 weeks | Chest pain Distant heart sounds Shock, rapid progression to cardiac arrest |
| Left ventricular aneurysm** | Up to several months | Heart failure Angina, ventricular arrhythmias |
*Usually due to right coronary artery occlusion.
**Usually due to left anterior descending artery occlusion
0–24 hours post-infarction
- Sudden cardiac death (SCD)
- MCC of out-of-hospital SCD in adults is ischemic heart disease.
- Strongest predictor of SCD post-MI: Severely decreased LVEF (≤ 35%).
- High-risk period: Greatest risk is within the first 30 days post-MI.
- Definition: A sudden death presumably caused by cardiac arrhythmia or hemodynamic catastrophe, which occurs either within an hour of symptom onset in patients with cardiovascular symptoms, or within 24 hours of being asymptomatic in patients with no cardiovascular symptoms.
- Pathophysiology: Fatal ventricular arrhythmia is considered to be the underlying mechanism of SCD.
- Underlying conditions
- Coronary artery disease: present in ∼ 70% of cases in adults over 35 years
- Dilated cardiomyopathy/hypertrophic cardiomyopathy
- Myocarditis
- Hereditary ion channelopathies (e.g., long QT syndrome, Brugada syndrome)
- Prevention: installation of the implantable cardioverter-defibrillator device
- Papillary Muscle Dysfunction (not papillary muscle rupture)
- Mechanism: Acute ischemia (transient) or infarction of the papillary muscle (without rupture).
- Risk: Posteromedial muscle (fed by RCA) is most susceptible due to single blood supply.
- Clinical: New onset Mitral Regurgitation (usually mild-to-moderate), worsening pulmonary edema. t
- Arrhythmias: a common cause of death in MI patients in the first 24 hours
- Immediate Phase (0–48 hours)
- Ventricular Fibrillation/Tachycardia (VF/VT):
- The most common cause of death in the immediate post-MI period.
- Due to acute electrical instability.
- Bradycardia & AV Block:
- Most common with Inferior MI (RCA occlusion affecting SA/AV nodes). c
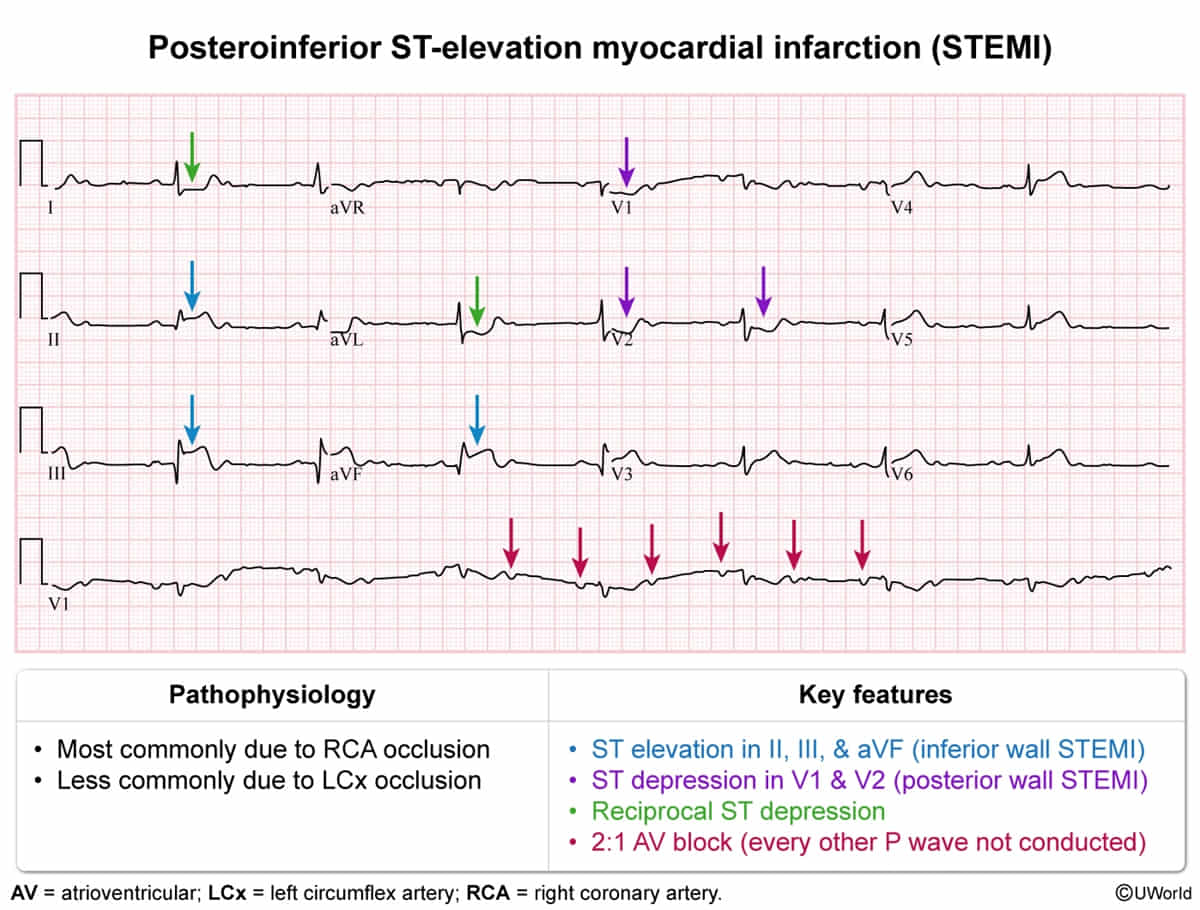
- Usually transient and resolves with reperfusion.
- AV block with Anterior MI (LAD occlusion) is less common but indicates extensive necrosis and carries a poor prognosis.
- Most common with Inferior MI (RCA occlusion affecting SA/AV nodes). c
- Accelerated Idioventricular Rhythm (AIVR):
- Regular, wide-complex rhythm (60-110 bpm).
- Generally benign and often considered a sign of successful reperfusion.
- Ventricular Fibrillation/Tachycardia (VF/VT):
- Subacute/Chronic Phase (>2 days)
- Atrial Fibrillation (AFib):
- The most common sustained arrhythmia post-MI.
- Caused by atrial ischemia, inflammation, or high LA pressure from LV dysfunction.
- Worsens prognosis (↑ risk of stroke, heart failure).
- Sustained Monomorphic VT:
- Caused by a stable re-entry circuit that forms around the healed infarct scar.
- A major cause of late sudden cardiac death.
- Atrial Fibrillation (AFib):
- Immediate Phase (0–48 hours)
1–3 days post-infarction
- Early infarct-associated pericarditis
- This pericarditis is a reaction to necrosis of the myocardium near the epicardial surface; usually localized to the areas overlying the necrotic myocardial segment.
- Clinical features of acute pericarditis, including:
- Friction rub
- Pleuritic chest pain
- Dry cough
- Due to inflammation of the mediastinal pleura adjoining the pericardium.
- Diffuse ST elevations on ECG
- Pericardial effusion
- Prognosis: usually self-limiting
Tip
- Early: peri-infarction pericarditis
- Late: Dressler syndrome
3–14 days post-infarction
- Papillary Muscle Rupture
- Risk Factors: Inferior MI (RCA occlusion). The posteromedial papillary muscle is highly vulnerable due to single blood supply (from PDA).
- Clinical Features:
- Acute, severe pulmonary edema / respiratory distress.
- Cardiogenic shock.
- Murmur: New-onset holosystolic murmur at the apex, radiating to axilla (Mitral Regurgitation). Note: Murmur may be soft if left atrial pressure rapidly equalizes.
- Diagnosis:
- Initial/Best: Transthoracic Echocardiogram (TTE) showing flail leaflet and severe MR.
- Management:
- Stabilize: Intra-aortic balloon pump (IABP) / inotropes for cardiogenic shock. Vasodilators (Nitroprusside) to ↓ afterload only if BP tolerates.
- Definitive: Emergent surgical valve repair/replacement.
- Interventricular Septal Rupture (Acquired VSD)
- Risk Factors: Anterior MI (LAD occlusion - apical septum) or Inferior MI (RCA occlusion - basal septum).
- Clinical Features:
- Acute biventricular failure / Cardiogenic shock.
- Murmur: New, harsh holosystolic murmur at Left Lower Sternal Border (LLSB).
- Palpable thrill frequently present.
- Diagnosis:
- Initial: TTE showing left-to-right shunt.
- Key Hemodynamics: Right heart catheterization shows an O2 saturation “step-up” from Right Atrium (RA) to Right Ventricle (RV).
- Management:
- Stabilize: IABP / inotropes.
- Definitive: Emergent surgical closure.
- Left Ventricular Free Wall Rupture
- Risk Factors: Anterior MI (LAD occlusion). More common in older females, first-time MI (lack of collateral circulation).
- Clinical Features:
- Sudden profound hypotension or Pulseless Electrical Activity (PEA) arrest.
- Cardiac Tamponade physiology: Beck’s Triad (JVD, muffled heart sounds, hypotension), pulsus paradoxus.
- Diagnosis:
- Initial: Bedside TTE (FAST scan equivalent) showing massive pericardial effusion with RV diastolic collapse.
- Management:
- Stabilize: Emergency pericardiocentesis (temporary bridge to relieve tamponade).
- Definitive: Emergent thoracotomy & surgical repair. Very high mortality.
- Contained ventricular rupture (pseudoaneurysm): refers to the outpouching of the ventricular wall rupture that is contained by either the pericardium, a thrombus, or scar tissue
- Complications
- Rupture → cardiac tamponade (risk of rupture is higher than in a true aneurysm)
- Associated with mural thromboembolism, decreased cardiac output, and increased risk of arrhythmia
- Complications
2 weeks to months post-infarction
Postmyocardial infarction syndrome (Dressler syndrome)
Pericarditis occurring 2–10 weeks post-MI without an infective cause
- Pathophysiology: thought to be due to circulating antibodies against cardiac muscle cells (autoimmune etiology) → immune complex deposition → inflammation
- Clinical features
- Signs of Pericarditis: pleuritic chest pain , dry cough , friction rub
- Fever
- Laboratory findings: leukocytosis, ↑ serum troponin levels
- ECG: diffuse ST elevations
- Treatment: NSAIDs (e.g., aspirin), colchicine
- Complications (rare): hemopericardium
Left Ventricular Aneurysm
- Pathophysiology: Thinning and fibrosis of the infarcted myocardium result in a true aneurysm (contains all 3 myocardial layers: endocardium, myocardium, epicardium). The fibrotic scar tissue creates a dyskinetic or akinetic wall segment that bulges during systole.
- While LV pseudoaneurysms (contained ruptures) have a high risk of rupture, true LV aneurysms are composed of dense, fibrotic scar tissue and rarely rupture c
- Clinical Features
- Often asymptomatic and discovered incidentally.
- Heart Failure (HF): Progressive dyspnea, orthopnea, edema (due to paradoxical wall motion decreasing LV ejection fraction).
- Arrhythmias: Palpitations, syncope (due to re-entrant ventricular tachycardia [VT] originating from the scar border).
- Systemic Embolization: Stroke, acute limb ischemia, or mesenteric ischemia (due to dislodgment of an LV mural thrombus).
- Physical Exam: Double or paradoxical apical impulse (PMI).
- Diagnosis
- Initial: ECG
- Classic finding: Persistent ST-segment elevation and deep Q waves in the leads of the previous MI (usually anterior leads V1-V4), weeks after the initial event. c
- Confirmatory/Gold Standard: Transthoracic Echocardiogram (TTE)
- Findings: Thin, dyskinetic, or akinetic LV wall with a wide neck and outward bulging during systole. May visualize a mural thrombus.
- Key Labs: BNP/NT-proBNP (elevated if HF present), Troponin (usually baseline, differentiating from re-infarction).
- Initial: ECG
Late Post-MI Arrhythmias
- Pathophysiology & Risk Factors
- Mechanism: Scar-mediated reentry. Fibrous tissue (macroscopic scar) replacing necrotic myocardium creates anatomic circuits for electrical reentry.
- Note: This contrasts with early post-MI arrhythmias (<48 hrs), which are due to acute ischemia, abnormal automaticity, and functional reentry.
- Most Common Arrhythmia: Monomorphic Ventricular Tachycardia (VT), which can rapidly degenerate into Ventricular Fibrillation (VF).
- Ventricular Aneurysm: A specific structural complication occurring weeks to months post-MI. The dyskinetic, scarred outpouching serves as a highly arrhythmogenic focus for refractory VT. c
- Clinical Features
- Palpitations, presyncope, or sudden syncope.
- Sudden Cardiac Death (SCD): Late post-MI VT/VF is the leading cause of SCD in ischemic cardiomyopathy.
- If Ventricular Aneurysm is present: Symptoms of decompensated HF (dyspnea, edema) or systemic emboli (stroke/TIA from mural thrombus).