Carcinogenesis
Carcinogens
- Chemical & Environmental Carcinogens
- Aflatoxins (from Aspergillus) → Hepatocellular carcinoma
- Associated with stored grains/nuts in humid climates.
- Mechanism: Induces p53 mutation (G:C → T:A transversion).
- Alkylating agents (e.g., chemotherapeutics like cyclophosphamide) → Leukemia/Lymphoma
- Common long-term side effect of chemo.
- Aromatic amines (e.g., aniline dyes, benzidine) → Transitional cell carcinoma of the bladder
- Classic Hx: Worker in textile or dye industry.
- Arsenic → Squamous cell carcinoma of the skin, Lung cancer, Angiosarcoma of the liver.
- Exposure: Herbicides, metal smelting, contaminated water.
- Asbestos → Bronchogenic carcinoma (most common malignancy), Mesothelioma (most specific).
- Classic Hx: Plumber, shipyard worker, insulation installer.
- Histology: Asbestos (ferruginous) bodies.
- Benzene → Acute myeloid leukemia (AML)
- Exposure: Industrial solvent, found in gasoline. Causes bone marrow suppression.
- Cigarette Smoke (2-Naphthylamine) → Transitional cell carcinoma (bladder), Squamous cell carcinoma (cervix, esophagus, larynx, lung), Small cell carcinoma (lung), Renal cell carcinoma, Pancreatic adenocarcinoma. t
- Polycyclic aromatic hydrocarbons are a key component.
- Ethanol → Squamous cell carcinoma (esophagus), Hepatocellular carcinoma.
- Nitrosamines (smoked foods) → Gastric adenocarcinoma (intestinal type).
- Radon → Lung cancer
- Second leading cause after smoking; risk is synergistic with smoking.
- Exposure: Decay of uranium in soil, accumulates in basements.
- Vinyl chloride → Angiosarcoma of the liver.
- Exposure: Used in making PVC pipes.
- Aflatoxins (from Aspergillus) → Hepatocellular carcinoma
- Oncogenic Viruses
- Epstein-Barr Virus (EBV) → Burkitt lymphoma, Hodgkin lymphoma (mixed cellularity subtype), Nasopharyngeal carcinoma, primary CNS lymphoma in immunocompromised pts.
- Hepatitis B Virus (HBV) & Hepatitis C Virus (HCV) → Hepatocellular carcinoma.
- Chronic inflammation → cirrhosis → carcinoma.
- Human Herpesvirus 8 (HHV-8) → Kaposi sarcoma.
- Classic settings: Elderly Eastern European men, AIDS pts, transplant recipients.
- Human Papillomavirus (HPV) → Squamous cell carcinoma (cervix, anus, vagina, vulva), Adenocarcinoma of the cervix, Oropharyngeal cancer.
- High-risk types: 16, 18.
- Mechanism: E6 gene product inhibits p53; E7 gene product inhibits Rb.
- Other Infectious Agents
- Helicobacter pylori → Gastric adenocarcinoma & MALT Lymphoma.
- Schistosoma haematobium → Squamous cell carcinoma of the bladder.
- Endemic to Middle East, Africa. Classic Hx: Male from Egypt with hematuria.
- Clonorchis sinensis (Chinese liver fluke) → Cholangiocarcinoma.
- Ingestion of undercooked fish.
- Radiation
- Ionizing radiation (e.g., nuclear accidents, radiotherapy) → Papillary carcinoma of the thyroid, Leukemias (AML, CML).
- Non-ionizing radiation (UVB sunlight) → Basal cell carcinoma, Squamous cell carcinoma, Melanoma of the skin.
- Mechanism: Forms pyrimidine dimers in DNA, normally repaired by nucleotide excision repair.
Defense mechanisms of malignant cells
I. Immune Evasion
- ↓ MHC Class I Expression: Prevents tumor antigen presentation to CD8+ cytotoxic T-cells, rendering the cancer cell “invisible.”
- Upregulation of Immune Checkpoints:
- PD-L1 (on tumor cell) binds PD-1 (on T-cell) → induces T-cell exhaustion/anergy. (Target of Pembrolizumab, Nivolumab).
- CTLA-4 (on T-cell) outcompetes CD28 for B7 (on APCs) → inhibits T-cell activation. (Target of Ipilimumab).
- Immunosuppressive Microenvironment:
- Secretion of TGF-β and IL-10 → inhibit T-cells and NK cells.
- Recruitment of regulatory immune cells: Tregs, Myeloid-Derived Suppressor Cells (MDSCs), and M2 Macrophages.
- Antigen Loss: Tumor cells lose expression of recognizable neoantigens through mutation, leading to immune escape.
II. Drug Resistance
- Increased Drug Efflux:
- Upregulation of P-glycoprotein (MDR1 gene product), an ATP-dependent pump that removes chemo drugs (e.g., Vinca alkaloids, taxanes) from the cell.
- Modification of Drug Targets:
- Gene Amplification: e.g., DHFR amplification confers resistance to Methotrexate.
- Mutation: e.g., Kinase domain mutations in CML confer resistance to Imatinib.
- Enhanced DNA Repair: Increased capacity to repair damage caused by alkylating agents or radiation.
- Inhibition of Apoptosis:
- Upregulation of anti-apoptotic proteins (e.g., Bcl-2).
- Inactivation of pro-apoptotic proteins (e.g., mutations in TP53).
- Drug Inactivation: Cancer cells produce enzymes that metabolize and inactivate drugs.
Metastasis
Mechanisms of metastasis
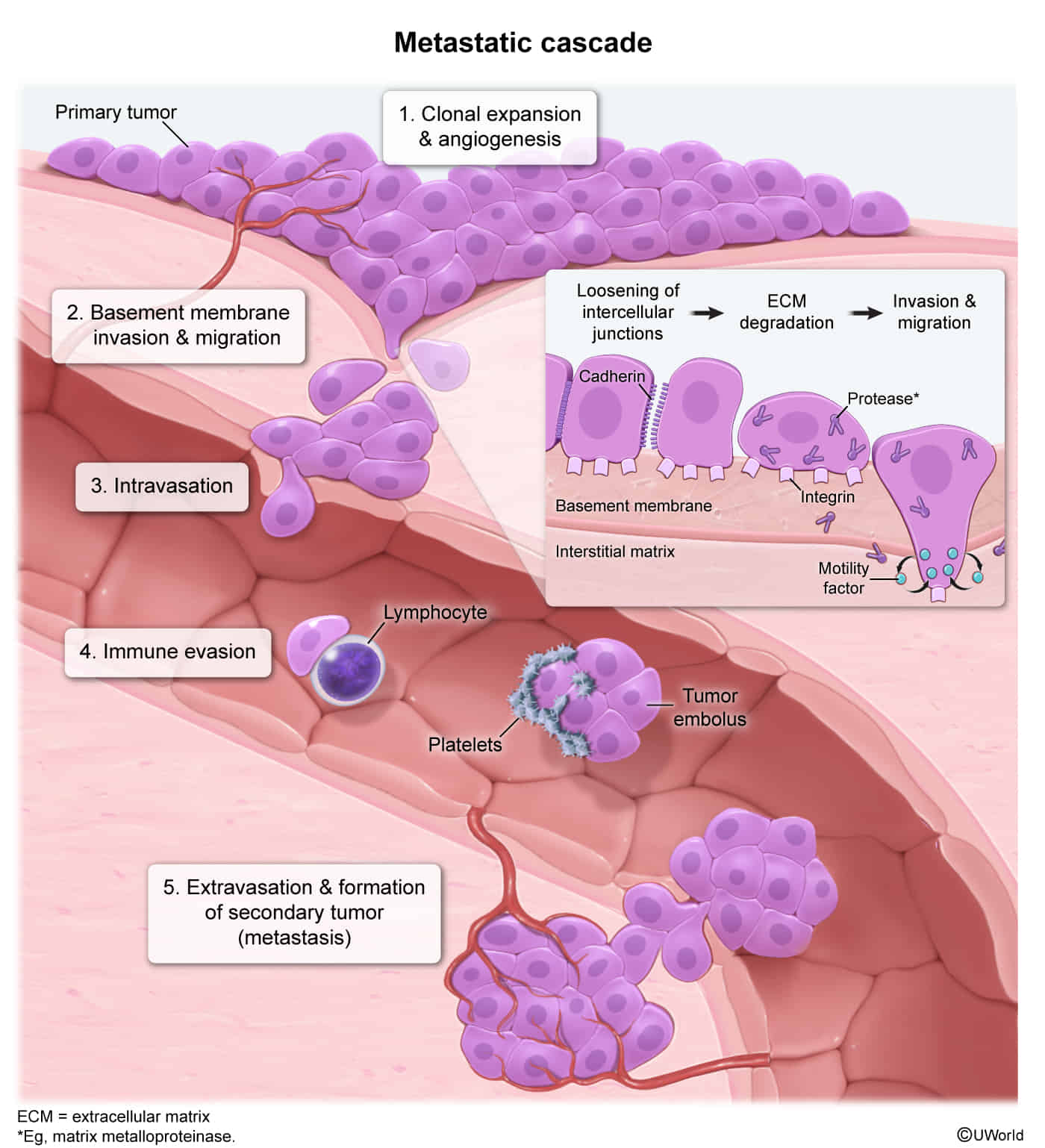
- 1. Invasion (Key Steps)
- Detachment: E-cadherin (loosens cell-cell adhesion).
- Attachment: Laminin receptors (binds to Basement Membrane).
- Degradation: Secretion of Metalloproteinases (MMPs) (specifically Type IV Collagenase) dissolves BM. t
- 2. Intravasation & Circulation
- Tumor cells enter vessels.
- Survival: Platelet aggregation coats tumor cells, protecting them from immune surveillance.
- 3. Angiogenesis
- Required for growth > 1–2 mm.
- Mediated by secretion of VEGF (Vascular Endothelial Growth Factor) and FGF (Fibroblast Growth Factor).
Types of metastasis
- Carcinomas → Primarily Lymphatic spread to regional lymph nodes.
- Sarcomas → Primarily Hematogenous spread.
A helpful mnemonic for these exceptions is “Four Rare Hematogenous Carcinomas”:
- Follicular Thyroid Carcinoma: Invades capsular blood vessels, a key feature distinguishing it from follicular adenoma.
- Renal Cell Carcinoma (RCC): Classically invades the renal vein and can extend as a tumor thrombus into the inferior vena cava (IVC) and even the right atrium.
- Hepatocellular Carcinoma (HCC): Frequently invades the portal and hepatic veins.
- Choriocarcinoma: A highly aggressive tumor of trophoblastic tissue that rapidly invades blood vessels, leading to early hematogenous spread to the lungs.